Energy Storage Science and Technology ›› 2024, Vol. 13 ›› Issue (12): 4368-4380.doi: 10.19799/j.cnki.2095-4239.2024.0731
• Special Issue on Thermochemical Energy Storage • Previous Articles Next Articles
Chao YU1,2( ), Gechuanqi PAN1(
), Gechuanqi PAN1( )
)
Received:2024-08-01
Revised:2024-10-23
Online:2024-12-28
Published:2024-12-23
Contact:
Gechuanqi PAN
E-mail:1240096418@qq.com;pangechuanqi@mail.163.com
CLC Number:
Chao YU, Gechuanqi PAN. Molecular dynamics study on structure and thermal properties of high-performance chloride molten salt[J]. Energy Storage Science and Technology, 2024, 13(12): 4368-4380.
Table 1
PRDF and CN characteristic of MgNaK eutectic salt vary with temperature"
| 不同配位 | T/K | rmax/Å | hmax | rmin/Å | N |
|---|---|---|---|---|---|
| Mg-Cl | 660 | 2.389 | 8.054 | 3.575 | 5.230 |
| 700 | 2.382 | 8.064 | 3.570 | 5.145 | |
| 800 | 2.371 | 8.041 | 3.534 | 4.958 | |
| 900 | 2.363 | 7.992 | 3.486 | 4.805 | |
| 1000 | 2.331 | 7.908 | 3.491 | 4.748 | |
| 1100 | 2.299 | 7.849 | 3.433 | 4.655 | |
| 1200 | 2.348 | 7.795 | 3.555 | 4.618 | |
| Mg-Mg | 660 | 3.652 | 1.671 | 5.401 | 3.075 |
| 700 | 3.652 | 1.605 | 5.303 | 2.895 | |
| 800 | 3.690 | 1.482 | 5.368 | 2.843 | |
| 900 | 3.708 | 1.389 | 5.332 | 2.701 | |
| 1000 | 3.718 | 1.330 | 5.258 | 2.608 | |
| 1100 | 3.665 | 1.292 | 5.174 | 2.500 | |
| 1200 | 3.773 | 1.263 | 5.335 | 2.482 | |
| Na-Cl | 660 | 2.787 | 4.193 | 4.010 | 6.400 |
| 700 | 2.783 | 4.109 | 3.997 | 6.287 | |
| 800 | 2.778 | 3.906 | 4.008 | 6.106 | |
| 900 | 2.764 | 3.753 | 4.045 | 6.000 | |
| 1000 | 2.721 | 3.621 | 4.098 | 6.047 | |
| 1100 | 2.679 | 3.509 | 3.998 | 5.760 | |
| 1200 | 2.736 | 3.425 | 4.079 | 5.573 | |
| K-Cl | 660 | 3.186 | 3.241 | 4.557 | 8.413 |
| 700 | 3.184 | 3.163 | 4.568 | 8.299 | |
| 800 | 3.169 | 2.988 | 4.592 | 8.060 | |
| 900 | 3.159 | 2.859 | 4.551 | 7.657 | |
| 1000 | 3.117 | 2.743 | 4.578 | 7.646 | |
| 1100 | 3.075 | 2.655 | 4.525 | 7.392 | |
| 1200 | 3.129 | 2.584 | 4.603 | 7.066 | |
| Cl-Cl | 660 | 3.621 | 2.548 | 5.304 | 12.342 |
| 700 | 3.626 | 2.472 | 5.314 | 12.219 | |
| 800 | 3.654 | 2.333 | 5.327 | 11.875 | |
| 900 | 3.665 | 2.233 | 5.327 | 11.505 | |
| 1000 | 3.639 | 2.153 | 5.353 | 11.597 | |
| 1100 | 3.613 | 2.089 | 5.358 | 11.588 | |
| 1200 | 3.708 | 2.036 | 5.439 | 11.009 | |
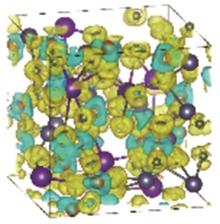
Fig.7
Charge density difference of the MgNaK eutectic salt at 1000 K. The yellow contours indicate electron accumulation, while the blue contours represent electron depletion. The contours are chosen to be 0.005 e/Å3. The Mg, Na, K and Cl are colored as orange, grey, purple and green, respectively"
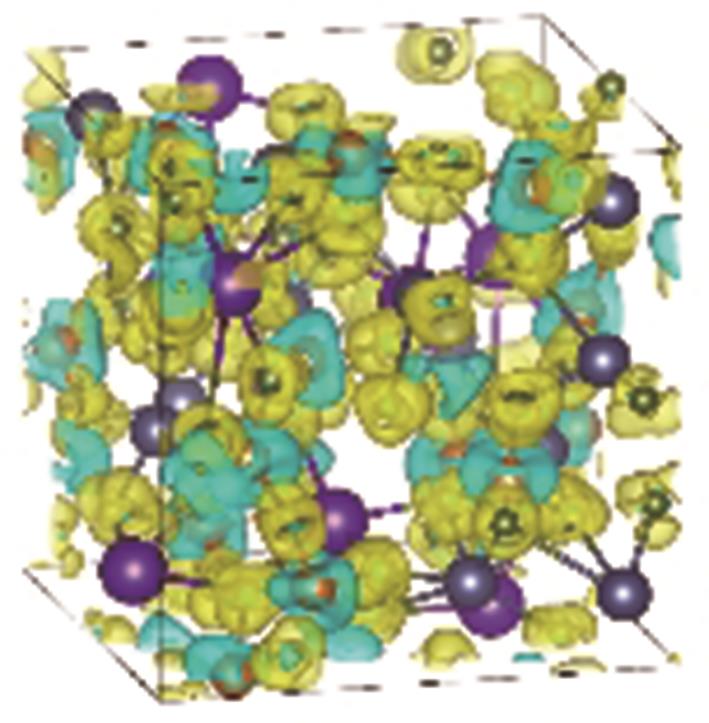
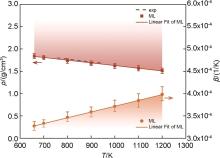
Fig.9
The density and constant pressure specific heat capacity of the MgNaK eutectic salt exhibit a dependence on temperature. The experimental values are represented by the dashed line, while the solid symbols and lines depict the simulated values obtained from the ML model and the linear fit, respectively"
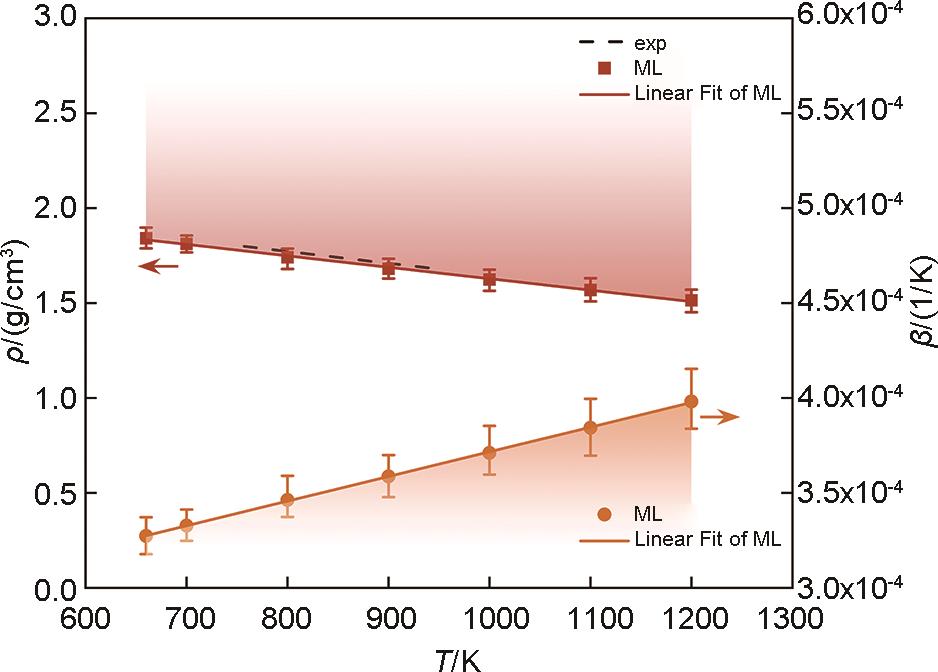

Fig.16
Temperature-dependent η of MgNaK eutectic salt (a) ηvs.T, (b) ln(ηT-1/2) vs. (T-T0)-1. Simulated values and VTF fit of ML are represented by solid red circles and lines, respectively. Simulated values of Villada and Li are denoted by solid green and blue squares. The open orange squares correspond to experimental values obtained in this study"

| 1 | NUNES V M B, QUEIRÓS C S, LOURENÇO M J V, et al. Molten salts as engineering fluids–A review Part I. Molten alkali nitrates[J]. Applied Energy, 2016, 183: 603-611. DOI: 10.1016/j. apenergy.2016.09.003. |
| 2 | 马广超, 狄跃忠, 彭建平, 等. 青海盐湖水氯镁石利用技术现状[J]. 矿产保护与利用, 2019, 39(3): 160-166. DOI: 10.13779/j.cnki.issn1001-0076.2019.03.025. |
| MA G C, DI Y Z, PENG J P, et al. Utilization technical status of bischofite in Qinghai salt lake[J]. Conservation and Utilization of Mineral Resources, 2019, 39(3): 160-166. DOI: 10.13779/j.cnki.issn1001-0076.2019.03.025. | |
| 3 | 吕秀梅, 尤静林, 王媛媛, 等. Na3AlF6-Al2O3系熔盐离子结构的拉曼光谱研究[J]. 光散射学报, 2015, 27(1): 39-43. DOI: 10.13883/j.issn1004-5929.201501009. |
| LV X M, YOU J L, WANG Y Y, et al. Raman spectroscopic study on the structure of Na3AlF6-Al2O3 molten salt system[J]. The Journal of Light Scattering, 2015, 27(1): 39-43. DOI: 10.13883/j.issn1004-5929.201501009. | |
| 4 | SMITH A L, VERLEG M N, VLIELAND J, et al. In situ high-temperature EXAFS measurements on radioactive and air-sensitive molten salt materials[J]. Journal of Synchrotron Radiation, 2019, 26(Pt 1): 124-136. DOI: 10.1107/S160057751801648X. |
| 5 | SUN J, GUO X J, ZHOU J, et al. Investigation of the local structure of molten ThF4-LiF and ThF4-LiF-BeF2 mixtures by high-temperature X-ray absorption spectroscopy and molecular-dynamics simulation[J]. Journal of Synchrotron Radiation, 2019, 26(Pt 5): 1733-1741. DOI: 10.1107/S1600577519009718. |
| 6 | GHERIBI A E, TORRES J A, CHARTRAND P. Recommended values for the thermal conductivity of molten salts between the melting and boiling points[J]. Solar Energy Materials and Solar Cells, 2014, 126: 11-25. DOI: 10.1016/j.solmat.2014.03.028. |
| 7 | AN X H, CHENG J H, YIN H Q, et al. Thermal conductivity of high temperature fluoride molten salt determined by laser flash technique[J]. International Journal of Heat and Mass Transfer, 2015, 90: 872-877. DOI: 10.1016/j.ijheatmasstransfer. 2015. 07.042. |
| 8 | WANG J, WU J, SUN Z, et al. Molecular dynamics study of the transport properties and local structures of molten binary systems (Li, Na)Cl, (Li, K)Cl and (Na, K)Cl[J]. Journal of Molecular Liquids, 2015, 209: 498-507. DOI: 10.1016/j.molliq.2015.06.021. |
| 9 | WANG J, WU J, LU G M, et al. Molecular dynamics study of the transport properties and local structures of molten alkali metal chlorides. Part III. Four binary systems LiCl-RbCl, LiCl-CsCl, NaCl-RbCl and NaCl-CsCl[J]. Journal of Molecular Liquids, 2017, 238: 236-247. DOI: 10.1016/j.molliq.2017.03.103. |
| 10 | WU J, WANG J, NI H O, et al. The influence of NaCl concentration on the (LiCl-KCl) eutectic system and temperature dependence of the ternary system[J]. Journal of Molecular Liquids, 2018, 253: 96-112. DOI: 10.1016/j.molliq.2017.11.068. |
| 11 | PAN G C, DING J, WANG W L, et al. Molecular simulations of the thermal and transport properties of alkali chloride salts for high-temperature thermal energy storage[J]. International Journal of Heat and Mass Transfer, 2016, 103: 417-427. DOI: 10.1016/j.ijheatmasstransfer.2016.07.042. |
| 12 | DING J, PAN G, DU L C, et al. Theoretical prediction of the local structures and transport properties of binary alkali chloride salts for concentrating solar power[J]. Nano Energy, 2017, 39: 380-389. DOI: 10.1016/j.nanoen.2017.07.020. |
| 13 | ISHII Y, KASAI S, SALANNE M, et al. Transport coefficients and the Stokes-Einstein relation in molten alkali halides with polarisable ion model[J]. Molecular Physics, 2015, 113(17/18): 2442-2450. DOI: 10.1080/00268976.2015.1046527. |
| 14 | CORRADINI D, MADDEN P A, SALANNE M. Coordination numbers and physical properties in molten salts and their mixtures[J]. Faraday Discussions, 2016, 190: 471-486. DOI: 10.1039/c5fd00223k. |
| 15 | WEN T Q, WANG C Z, KRAMER M J, et al. Development of a deep machine learning interatomic potential for metalloid-containing Pd-Si compounds[J]. Physical Review B, 2019, 100(17): 174101. DOI: 10.1103/physrevb.100.174101. |
| 16 | PAN G, CHEN P, YAN H, et al. A DFT accurate machine learning description of molten ZnCl2 and its mixtures: 1. Potential development and properties prediction of molten ZnCl2[J]. Computational Materials Science, 2020, 185: 109955. DOI: 10.1016/j.commatsci.2020.109955. |
| 17 | PAN G, DING J, DU Y F, et al. A DFT accurate machine learning description of molten ZnCl2 and its mixtures: 2. Potential development and properties prediction of ZnCl2-NaCl-KCl ternary salt for CSP[J]. Computational Materials Science, 2021, 187: 110055. DOI: 10.1016/j.commatsci.2020.110055. |
| 18 | GRIMME S. Accurate description of van der Waals complexes by density functional theory including empirical corrections[J]. Journal of Computational Chemistry, 2004, 25(12): 1463-1473. DOI: 10.1002/jcc.20078. |
| 19 | MARTÍNEZ L, ANDRADE R, BIRGIN E G, et al. PACKMOL: A package for building initial configurations for molecular dynamics simulations[J]. Journal of Computational Chemistry, 2009, 30(13): 2157-2164. DOI: 10.1002/jcc.21224. |
| 20 | LI X J, LI N, LIU W H, et al. Unrevealing the thermophysical properties and microstructural evolution of MgCl2-NaCl-KCl eutectic: FPMD simulations and experimental measurements[J]. Solar Energy Materials and Solar Cells, 2020, 210: 110504. DOI: 10.1016/j.solmat.2020.110504. |
| 21 | WANG H, ZHANG L F, HAN J Q, et al. DeePMD-kit: A deep learning package for many-body potential energy representation and molecular dynamics[J]. Computer Physics Communications, 2018, 228: 178-184. DOI: 10.1016/j.cpc.2018.03.016. |
| 22 | PLIMPTON S. Fast parallel algorithms for short-range molecular dynamics[J]. Journal of Computational Physics, 1995, 117(1): 1-19. DOI: 10.1006/jcph.1995.1039. |
| 23 | SHARMA B K, WILSON M. Intermediate-range order in molten network-forming systems[J]. Physical Review B, 2006, 73(6): 060201. DOI: 10.1103/physrevb.73.060201. |
| 24 | MAINTZ S, DERINGER V L, TCHOUGRÉEFF A L, et al. LOBSTER: A tool to extract chemical bonding from plane-wave based DFT[J]. Journal of Computational Chemistry, 2016, 37(11): 1030-1035. DOI: 10.1002/jcc.24300. |
| 25 | LI Y Y, XU X K, WANG X X, et al. Survey and evaluation of equations for thermophysical properties of binary/ternary eutectic salts from NaCl, KCl, MgCl2, CaCl2, ZnCl2 for heat transfer and thermal storage fluids in CSP[J]. Solar Energy, 2017, 152: 57-79. DOI: 10.1016/j.solener.2017.03.019. |
| 26 | LIANG W S, LU G M, YU J G. Molecular dynamics simulations of molten magnesium chloride using machine-learning-based deep potential[J]. Advanced Theory and Simulations, 2020, 3(12): DOI: 10.1002/adts.202000180. |
| 27 | PAN G, DING J, CHEN P, et al. Finite-size effects on thermal property predictions of molten salts[J]. Solar Energy Materials and Solar Cells, 2021, 221: 110884. DOI: 10.1016/j.solmat. 2020. 110884. |
| 28 | VILLADA C, DING W J, BONK A, et al. Engineering molten MgCl2-KCl-NaCl salt for high-temperature thermal energy storage: Review on salt properties and corrosion control strategies[J]. Solar Energy Materials and Solar Cells, 2021, 232: 111344. DOI: 10.1016/j.solmat.2021.111344. |
| 29 | PAN G, WEI X L, YU C, et al. Thermal performance of a binary carbonate molten eutectic salt for high-temperature energy storage applications[J]. Applied Energy, 2020, 262: 114418. DOI: 10.1016/j.apenergy.2019.114418. |
| [1] | Yuhang YUAN, Yuchen GAO, Jundong ZHANG, Yanbin GAO, Chaolong WANG, Xiang CHEN, Qiang ZHANG. The application of large language models in energy storage research [J]. Energy Storage Science and Technology, 2024, 13(9): 2907-2919. |
| [2] | Wentao ZHU, Yang ZHOU, Yimin XU, Tao SHI. Application and optimization of battery energy storage technology in new energy generation system [J]. Energy Storage Science and Technology, 2024, 13(8): 2737-2739. |
| [3] | Chu ZHANG, Dongcai CHEN, Xiangping CHEN, Yongxiang CAI. Economic benefit analysis of optimal allocation of energy storage in multiple application scenarios [J]. Energy Storage Science and Technology, 2024, 13(6): 2078-2088. |
| [4] | Jian SUN, Jianlong TAO, Yunrong HU, Xiaolong CAO, Yongping YANG. Summary of research on power storage technology based on heat pump at home and abroad [J]. Energy Storage Science and Technology, 2024, 13(6): 1963-1976. |
| [5] | Li ZHOU, Yan LIU. Application and development of alloy materials in energy storage technology [J]. Energy Storage Science and Technology, 2024, 13(6): 1874-1876. |
| [6] | Zhenxin SUN, Zhiming ZHANG, Fubo MA, Congjin JIANG, Haoyi DU, Huanjun CHEN, Yukui ZHANG. Investigation of energy regulation performance based on entropy theory [J]. Energy Storage Science and Technology, 2024, 13(5): 1584-1591. |
| [7] | Tianchen LI, Jianzheng YIN, Dawei ZHANG, Xiaoheng LIU. Research on renewable energy grid integration strategy based on hydropower station energy storage technology [J]. Energy Storage Science and Technology, 2024, 13(2): 677-679. |
| [8] | Yujie ZHANG, Jiangyun CHEN, Jianqiang LI, Yanjun DAI. China Thermal Energy Storage Industry Development Report (2024)—Industry technologies, development status, and model projects [J]. Energy Storage Science and Technology, 2024, 13(12): 4452-4463. |
| [9] | Suping LIU. Analysis and practical application of energy storage technology of flow redox cells [J]. Energy Storage Science and Technology, 2024, 13(11): 3871-3873. |
| [10] | Qian LYU. Research on the application of virtual energy storage technology in power plants [J]. Energy Storage Science and Technology, 2024, 13(11): 4059-4061. |
| [11] | Su YAN, Fangfang ZHONG, Junwei LIU, Mei DING, Chuankun JIA. Key materials and advanced characterization of high-energy-density flow battery [J]. Energy Storage Science and Technology, 2024, 13(1): 143-156. |
| [12] | Jiajun ZHANG, Xiaoqiong LI, Zhentao ZHANG, Jiahao HAO, Pingyang ZHENG, Ze YU, Junling YANG, Yanan JING, Yunkai YUE. Research progress of compressed carbon dioxide energy storage system [J]. Energy Storage Science and Technology, 2023, 12(6): 1928-1945. |
| [13] | Tingting QIN, Xuezhi ZHOU, Dingzhang GUO, Yong SHENG, Yujie XU, Zhitao ZUO, Hui LI, Haisheng CHEN. Study on factors influencing rail gravity energy storage system efficiency [J]. Energy Storage Science and Technology, 2023, 12(3): 835-845. |
| [14] | Zhiwei ZHAO, Zhi YANG, Zhangquan PENG. Application of time-of-flight secondary ion mass spectrometry in lithium-based rechargeable batteries [J]. Energy Storage Science and Technology, 2022, 11(3): 781-794. |
| [15] | Liangbo QIAO, Xiaohu ZHANG, Xianzhong SUN, Xiong ZHANG, Yanwei MA. Advances in battery-supercapacitor hybrid energy storage system [J]. Energy Storage Science and Technology, 2022, 11(1): 98-106. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||
