Energy Storage Science and Technology ›› 2024, Vol. 13 ›› Issue (7): 2151-2160.doi: 10.19799/j.cnki.2095-4239.2024.0383
• Special Issue on Low Temperature Batteries • Previous Articles Next Articles
Jiaqi HUANG1( ), Jieming XIONG1, Enzhong TAN2(
), Jieming XIONG1, Enzhong TAN2( ), Xinyu SUN3, Liwei CHENG3, Hua WANG3(
), Xinyu SUN3, Liwei CHENG3, Hua WANG3( )
)
Received:2024-05-06
Revised:2024-05-23
Online:2024-07-28
Published:2024-07-23
Contact:
Enzhong TAN, Hua WANG
E-mail:2023540084@bipt.edu.cn;0020020500@bipt.edu.cn;wanghua8651@buaa.edu.cn
CLC Number:
Jiaqi HUANG, Jieming XIONG, Enzhong TAN, Xinyu SUN, Liwei CHENG, Hua WANG. Revisiting the Na metal half-cell at low-temperature[J]. Energy Storage Science and Technology, 2024, 13(7): 2151-2160.

Fig. 1
(a) charge/discharge curves of Na||HC half-cells at 25 ℃ and -20 ℃; (b) Schematic diagram of the three-electrode cell; (c) Charge/discharge curves of Na||HC half-cell (up) and the corresponding potential variation of Na counter electrode (down), inset is the digital photo of the three-electrode cell"
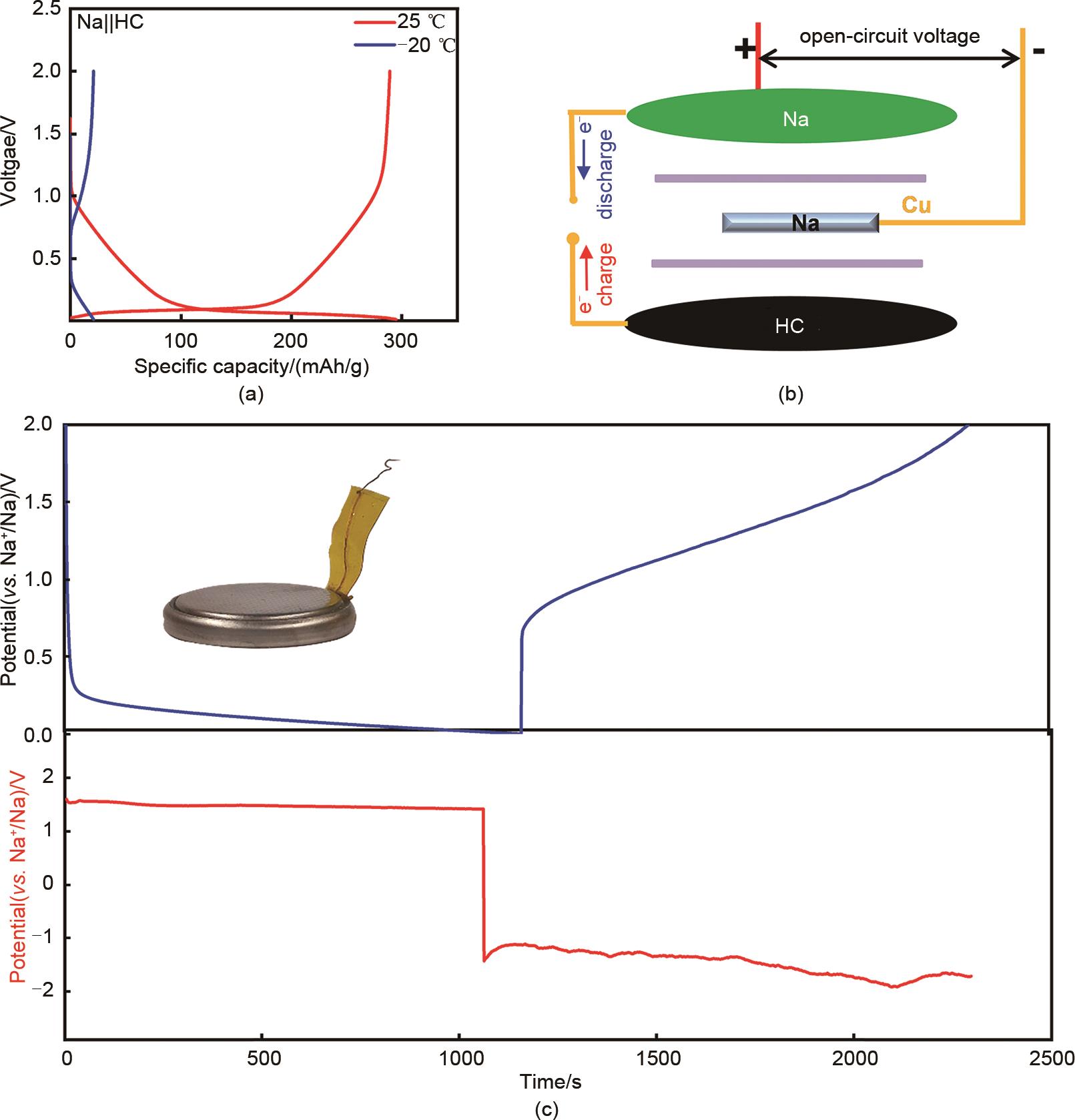

Fig. 5
(a) Voltage of Na15Sn4@Na vs. Na+/Na; (b) Cycling performance of Na15Sn4@Na||Na15Sn4@Na cell at -20 ℃; (c) Charge/discharge curves of Na15Sn4@Na||Na15Sn4@Na and Na||Na symmetric-cell at -20 ℃; (d) Nyquist curves of Na15Sn4||Na15Sn4 and Na||Na symmetric-cell at (e) 25 and -20 ℃; (f) Impedance comparison of Na15Sn4@Na||Na15Sn4@Na and Na||Na cells at 25 and -20 ℃"
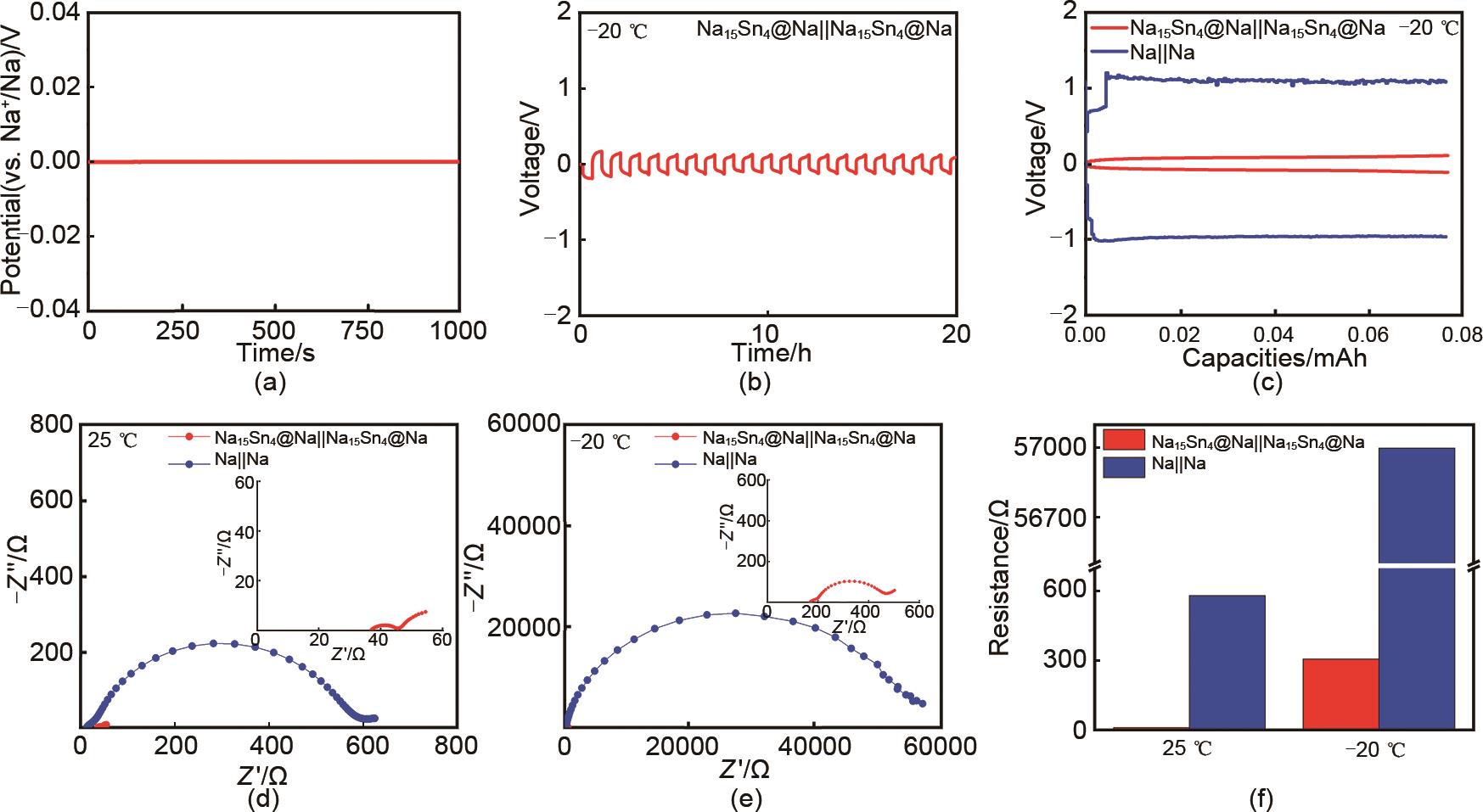
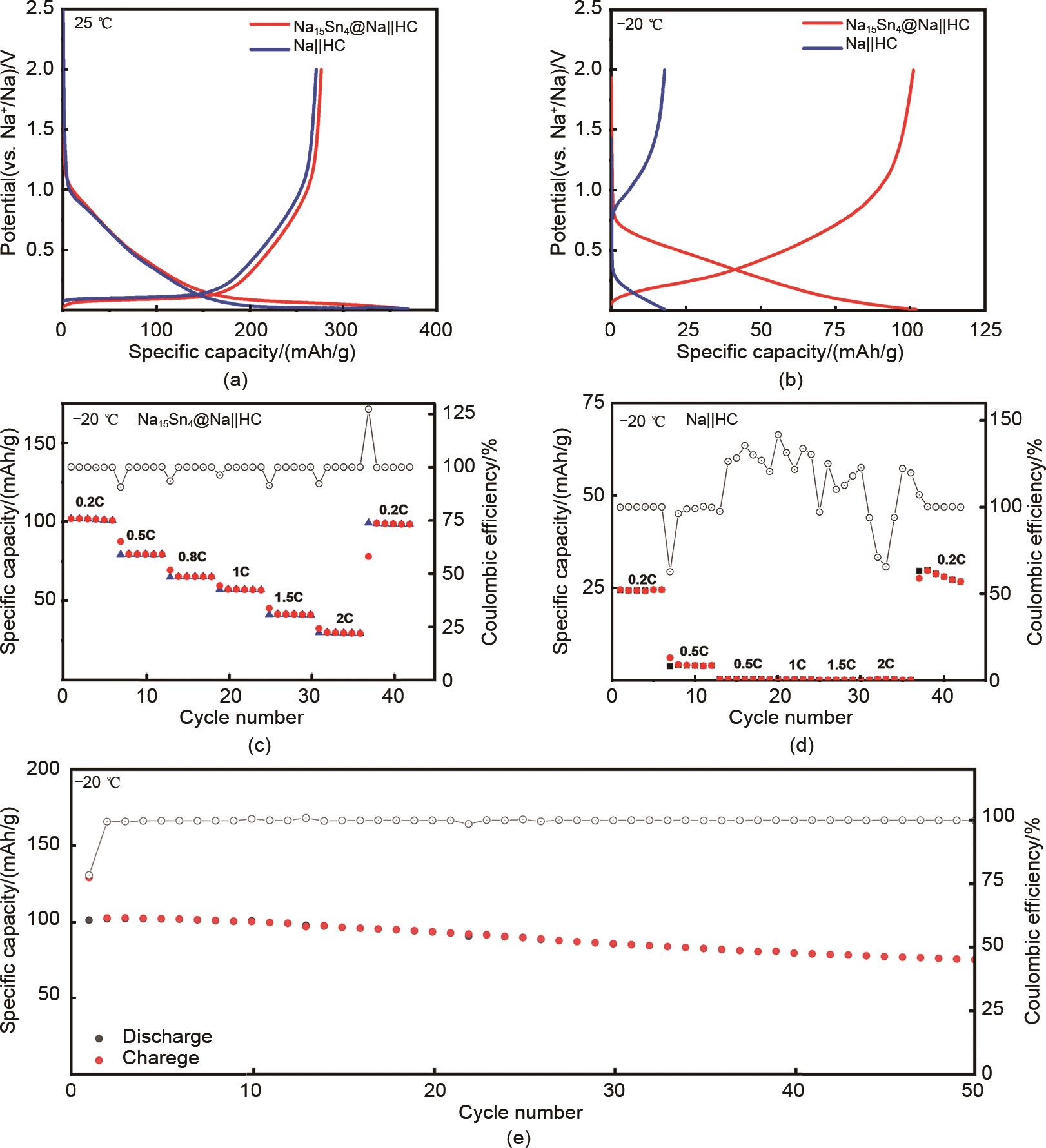
Fig. 7
(a) Initial charge/discharge curves of Na15Sn4@Na||HC and Na||HC half-cells at 25℃; (b) Charge/discharge curves of Na15Sn4@Na||HC and Na||HC half-cells at -20 ℃; (c) Rate performance of Na15Sn4@Na||HC half cells at -20 ℃; (d) Rate performance of Na||HC half cells at -20 ℃; (e) Cycling performance of Na15Sn4@Na||HC half-cell at -20 ℃"
| 31 | SHI P, HOU L P, JIN C B, et al. A successive conversion-deintercalation delithiation mechanism for practical composite lithium anodes[J]. Journal of the American Chemical Society, 2022, 144(1): 212-218. |
| 32 | CHENG L W, LAN H, GAO Y, et al. Realizing low-temperature graphite-based rechargeable potassium-ion full battery[J]. Angewandte Chemie International Edition, 2024, 63(7): 2315624. |
| 33 | WANG Y Y, LAN H, DONG S, et al. A high-power rechargeable sodium-ion full battery operating at -40 ℃[J]. Advanced Functional Materials, 2024: 2315498. |
| 34 | YAMAMOTO T, NOHIRA T, HAGIWARA R, et al. Thermodynamic studies on Sn-Na alloy in an intermediate temperature ionic liquid NaFSA-KFSA at 363K[J]. Journal of Power Sources, 2013, 237: 98-103. |
| 35 | ZHANG B, ROUSSE G, FOIX D, et al. Microsized Sn as advanced anodes in glyme-based electrolyte for Na-ion batteries[J]. Advanced Materials, 2016, 28(44): 9824-9830. |
| 36 | CHEN M H, CHAO D L, LIU J L, et al. Rapid pseudocapacitive sodium-ion response induced by 2D ultrathin tin monoxide nanoarrays[J]. Advanced Functional Materials, 2017, 27(12): doi: 10.1002/adfm.201606232. |
| 37 | 陈宏善, 牛建中, 夏春谷, 等. 甲烷氧化偶联Na-W-Mn/SiO2催化剂的喇曼光谱[J]. 物理化学学报, 2000, 16(6): 543-546. |
| CHEN H S, NIU J Z, XIA C G, et al. Raman spectroscopy characterization of Na-W-Mn/SiO2 catalyst for oxidative coupling of methane[J]. Acta Physico-Chimica Sinica, 2000, 16(6): 543-546. | |
| 38 | DAHUNSI O J, LI B M, GAO S Y, et al. One-step synthesis of Na-Sn alloy with internal 3D Na15Sn4 support for fast and stable Na metal batteries[J]. ACS Applied Energy Materials, 2022, 5(1): 20-26. |
| 1 | 成伟翔, 黄兴文, 李越珠, 等. 层状金属二硫化物作为钠离子电池负极的研究进展[J]. 储能科学与技术, 2022, 11(10): 3062-3075. |
| CHENG W X, HUANG X W, LI Y Z, et al. Advances in layered metal disulfide as anode material for Na-ion batteries[J]. Energy Storage Science and Technology, 2022, 11(10): 3062-3075. | |
| 2 | WANG Y Y, HOU B H, GUO J Z, et al. An ultralong lifespan and low-temperature workable sodium-ion full battery for stationary energy storage[J]. Advanced Energy Materials, 2018, 8(18): 1703252. |
| 3 | KIM E Y, MOHAMMADIROUDBARI M, CHEN F, et al. A carbonyl and azo-based polymer cathode for low-temperature Na-ion batteries[J]. ACS Nano, 2024, 18(5): 4159-4169. |
| 4 | 陈珂君, 范利君. 钴掺杂FeS2的可控制备及储钠特性研究[J]. 储能科学与技术, 2023, 12(10): 3056-3063. |
| CHEN K J, FAN L J. Controllable synthesis of Co2+-doped FeS2 and their sodium storage performances[J]. Energy Storage Science and Technology, 2023, 12(10): 3056-3063. | |
| 5 | WANG H, YU D D, KUANG C W, et al. Alkali metal anodes for rechargeable batteries[J]. Chem, 2019, 5(2): 313-338. |
| 6 | LI Y Q, YANG Y, LU Y X, et al. Ultralow-concentration electrolyte for Na-ion batteries[J]. ACS Energy Letters, 2020, 5(4): 1156-1158. |
| 7 | FAN X L, JI X, CHEN L, et al. All-temperature batteries enabled by fluorinated electrolytes with non-polar solvents[J]. Nature Energy, 2019, 4: 882-890. |
| 8 | 梁君飞, 李艳梅, 袁浩, 等. 低温锂离子电池研究进展[J]. 北京航空航天大学学报, 2021, 47(11): 2155-2174. |
| LIANG J F, LI Y M, YUAN H, et al. Research progress of low-temperature lithium-ion battery[J]. Journal of Beijing University of Aeronautics and Astronautics, 2021, 47(11): 2155-2174. | |
| 9 | DENG T, JI X, ZOU L F, et al. Interfacial-engineering-enabled practical low-temperature sodium metal battery[J]. Nature Nanotechnology, 2022, 17: 269-277. |
| 10 | ZHANG W L, LU Y, WAN L, et al. Engineering a passivating electric double layer for high performance lithium metal batteries[J]. Nature Communications, 2022, 13: 2029. |
| 11 | FANG H Y, HUANG Y H, HU W, et al. Regulating ion-dipole interactions in weakly solvating electrolyte towards ultra-low temperature sodium-ion batteries[J]. Angewandte Chemie, 2024, 136(15): e202400539. |
| 12 | TAN L, HU R Z, ZHANG H Y, et al. Subzero temperature promotes stable lithium storage in SnO2[J]. Energy Storage Materials, 2021, 36: 242-250. |
| 13 | ZHU Q N, YU D D, CHEN J C, et al. A 110 Wh·kg-1 Ah-level anode-free sodium battery at -40 ℃[J]. Joule, 8(21): 482-495. |
| 14 | ZHAO Y, ADAIR K R, SUN X L. Recent developments and insights into the understanding of Na metal anodes for Na-metal batteries[J]. Energy & Environmental Science, 2018, 11(10): 2673-2695.[LinkOut] |
| 15 | XIAO Y, XU R, YAN C, et al. A toolbox of reference electrodes for lithium batteries[J]. Advanced Functional Materials, 2022, 32(13): 2108449. |
| 16 | TANG Z, WANG H, WU P F, et al. Electrode-electrolyte interfacial chemistry modulation for ultra-high rate sodium-ion batteries[J]. Angewandte Chemie (International Ed in English), 2022, 61(18): e202200475. |
| 17 | WANG J K, LIU J H, WANG L, et al. The significance of imperceptible current flowing through the lithium reference electrode in lithium ion batteries[J]. Journal of Power Sources, 2022, 546: 231953. |
| 18 | 余永诗, 夏先明, 黄弘扬, 等. 钠金属负极人工界面保护层的研究进展[J]. 储能科学与技术, 2023, 12(5): 1380-1391. |
| YU Y S, XIA X M, HUANG H Y, et al. Research progress on sodium metal anode modified by artificial interface layer[J]. Energy Storage Science and Technology, 2023, 12(5): 1380-1391. | |
| 19 | XIA X M, XU S T, TANG F, et al. A multifunctional interphase layer enabling superior sodium-metal batteries under ambient temperature and -40 ℃[J]. Advanced Materials, 2023, 35(11): 2209511. |
| 20 | ZHENG X Y, GU Z Y, FU J, et al. Knocking down the kinetic barriers towards fast-charging and low-temperature sodium metal batteries[J]. Energy & Environmental Science, 2021, 14(9): 4936-4947. |
| 21 | WANG Z Q, ZHENG X Y, LIU X Y, et al. Promoting fast Na ion transport at low temperatures for sodium metal batteries[J]. ACS Applied Materials & Interfaces, 2022, 14(36): 40985-40991. |
| 22 | YAN L, ZHANG G F, WANG J, et al. Revisiting electrolyte kinetics differences in sodium ion battery: Are esters really inferior to ethers?[J]. ENERGY & ENVIRONMENTAL MATERIALS, 2023, 6(4): 12523. |
| 23 | XU Y H, ZHU Y J, LIU Y H, et al. Electrochemical performance of porous carbon/tin composite anodes for sodium-ion and lithium-ion batteries[J]. Advanced Energy Materials, 2013, 3(1): 128-133. |
| 24 | SHENG M H, ZHANG F, JI B F, et al. A novel tin-graphite dual-ion battery based on sodium-ion electrolyte with high energy density[J]. Advanced Energy Materials, 2017, 7(7): 1601963. |
| 25 | YANG B, WANG J, ZHU Y Y, et al. Engineering hard carbon with high initial coulomb efficiency for practical sodium-ion batteries[J]. Journal of Power Sources, 2021, 492: 229656. |
| 26 | XIE L J, TANG C, BI Z H, et al. Hard carbon anodes for next-generation Li-ion batteries: Review and perspective[J]. Advanced Energy Materials, 2021, 11(38): 2101650. |
| 27 | 江成凡, 黄俊, 谢海波. 提高硬碳材料钠离子电池首次库仑效率的研究进展[J]. 储能科学与技术, 2024, 13(3): 825-840. |
| JIANG C F, HUANG J, XIE H B. Improving the initial coulombic efficiency of hard carbon materials for sodium-ion batteries[J]. Energy Storage Science and Technology, 2024, 13(3): 825-840. | |
| 28 | CHEN X Y, LIU C Y, FANG Y J, et al. Understanding of the sodium storage mechanism in hard carbon anodes[J]. Carbon Energy, 2022, 4(6): 1133-1150. |
| 29 | RUDOLA A, WRIGHT C J, BARKER J. Communication-surprisingly high fast charge volumetric capacities of hard carbon electrodes in sodium-ion batteries[J]. Journal of the Electrochemical Society, 2021, 168(11): 110534. |
| 30 | YAO Y X, CHEN X, YAO N, et al. Unlocking charge transfer limitations for extreme fast charging of Li-ion batteries[J]. Angewandte Chemie (International Ed in English), 2023, 62(4): e202214828. |
| [1] | Changhao LI, Shuping WANG, Xiankun YANG, Ziqi ZENG, Xinyue ZHOU, Jia XIE. Nonaqueous electrolyte in low-temperature lithium-ion battery [J]. Energy Storage Science and Technology, 2024, 13(7): 2286-2299. |
| [2] | Shijie LIAO, Ying WEI, Yunhui HUANG, Renzong HU, Henghui XU. 1,3-Difluorobenzene diluent-stabilizing electrode interface for high-performance low-temperature lithium metal batteries [J]. Energy Storage Science and Technology, 2024, 13(7): 2124-2130. |
| [3] | Xiang LI, Dezhong LIU, Kai YUAN, Dapeng CHEN. Solid-state electrolyte for low-temperature lithium metal batteries [J]. Energy Storage Science and Technology, 2024, 13(7): 2327-2347. |
| [4] | Xiongwen XU, Ying MO, Wang ZHOU, Huandong YAO, Juan HONG, Hua LEI, Jian TU, Jilei LIU. Effect of hard carbon kinetic properties on low-temperature performance of Na-ion batteries [J]. Energy Storage Science and Technology, 2024, 13(7): 2141-2150. |
| [5] | Weiqi LIN, Qiaoyu LU, Yuhong CHEN, Linyuan QIU, Yurong JI, Lianyu GUAN, Xiang DING. Advances in cathode materials for low-temperature sodium-ion batteries [J]. Energy Storage Science and Technology, 2024, 13(7): 2348-2360. |
| [6] | Zheng LI, Zhenzhong YANG, Qiong WANG, Renzong HU. Patent intelligence analysis of the research progress in low-temperature electrolytes for Li-ion batteries [J]. Energy Storage Science and Technology, 2024, 13(7): 2317-2326. |
| [7] | Lifeng WANG, Naiqing REN, Hai YANG, Yu YAO, Yan YU. Advances in low-temperature electrolytes for sodium-ion batteries [J]. Energy Storage Science and Technology, 2024, 13(7): 2206-2223. |
| [8] | Yuhao WANG, Zhiyong LI, Xin GUO. Applications and challenges of polymer-based electrolytes in low-temperature solid-state lithium batteries [J]. Energy Storage Science and Technology, 2024, 13(7): 2243-2258. |
| [9] | Zeheng LI, Lei XU, Yuxing YAO, Chong YAN, Ximin ZHAI, Xuechun HAO, Aibing CHEN, Jiaqi HUANG, Xiaofei BIE, Huanli SUN, Lizhen FAN, Qiang ZHANG. A review of electrolyte reducing lithium plating in low-temperature lithium-ion batteries [J]. Energy Storage Science and Technology, 2024, 13(7): 2192-2205. |
| [10] | Shuping WANG, Xiankun YANG, Changhao LI, Ziqi ZENG, Yifeng CHENG, Jia XIE. Diethyl ethylphosphonate-based flame-retardant wide-temperature-range electrolyte in lithium-ion batteries [J]. Energy Storage Science and Technology, 2024, 13(7): 2161-2170. |
| [11] | Lulu NIE, Lige YUAN. Application and optimization of intelligent electronic control system in low temperature battery management [J]. Energy Storage Science and Technology, 2024, 13(7): 2432-2434. |
| [12] | Hui FANG. Functional safety guarantee strategy for low temperature lithium battery energy storage system under network analysis mode [J]. Energy Storage Science and Technology, 2024, 13(7): 2447-2449. |
| [13] | Junjie LU, Dan PENG, Wenjing NI, Yuan YANG, Jinglun WANG. Research progress on electrolyte for Li/CF x battery [J]. Energy Storage Science and Technology, 2024, 13(5): 1487-1495. |
| [14] | Junli GUO. Legal governance measures for fire safety of electrochemical energy storage power stations [J]. Energy Storage Science and Technology, 2024, 13(5): 1744-1747. |
| [15] | Qianqian ZHANG. Application of phase change storage technology in food cold chain logistics [J]. Energy Storage Science and Technology, 2024, 13(2): 480-482. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||
